免疫チェックポイント阻害薬への期待と課題
Pharma Medica 37(1):75-79, 2019
はじめに
卵巣は後腹膜臓器であるため卵巣癌は発見が困難で,また,早期には無症状で経過することが少なくないため難治となることが多い。卵巣癌全体の6~7割はIII~IV期の進行癌である。組織型別では,明細胞腺癌および粘液性腺癌は,漿液性腺癌や類内膜腺癌に比べ,化学療法に対する感受性が低く,わが国では粘液性腺癌,明細胞腺癌の頻度が欧米に比べ高いという特徴がある1)。
また,化学療法に感受性である漿液性腺癌においても再発率は低くないことも今後の課題である。
そこで本稿では,卵巣癌初回化学療法の現状,免疫チェックポイント阻害薬を含む近年の薬剤の動向について紹介する。
卵巣癌初回化学療法の現状
卵巣癌の初回化学療法では,パクリタキセル+カルボプラチン併用療法であるconventional TC(c-TC)療法が世界的に標準療法とされている。このc-TC療法と比べて生存期間を延長するレジメンの1つとして,c-TC療法におけるパクリタキセルの投与間隔(3または4週に1回)を週1回に短縮したdose-dense TC(dd-TC)療法が知られている2)。
dd-TC療法については,わが国の婦人科悪性腫瘍研究機構(JGOG)が,Stage II~IVの上皮性卵巣癌,腹膜癌,卵管癌の患者631例を対象として,dd-TC療法とc-TC療法の有用性を比較した第III相試験であるJGOG3016試験を実施した3)。その結果,観察期間中央値29ヵ月時点での無増悪生存期間(PFS)中央値は,dd-TC療法群28.0ヵ月,c-TC療法群17.2ヵ月となり,c-TC療法群に比べdd-TC群で有意な延長が認められた〔ハザード比(HR)=0.71〕。さらに,フォローアップデータにおいて4),経過観察中央値6.4年時点のPFS中央値はdd-TC療法群28.2ヵ月,c-TC療法群17.5ヵ月となり,c-TC療法群に比べdd-TC群で有意な延長が認められた(HR=0.76)。全生存期間(OS)中央値はそれぞれ100.5ヵ月,62.2ヵ月となり,c-TC療法群に比べdd-TC群で延長が認められた(HR=0.79,図1)。
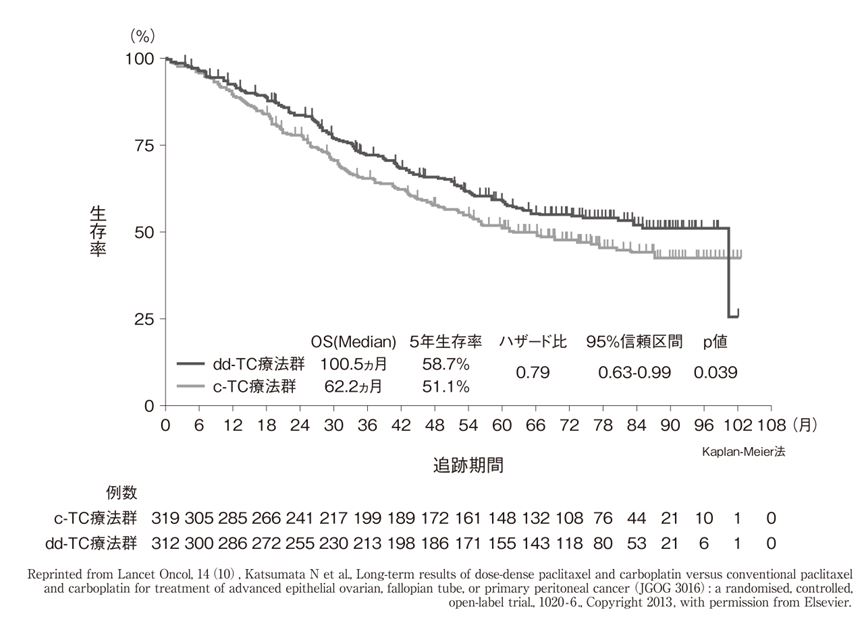
図1 JGOG3016試験:OS
本試験の結果などを踏まえ,わが国の卵巣がん治療ガイドラインでは,卵巣癌の初回化学療法として,c-TC療法は推奨グレードA,dd-TC療法は推奨グレードBに位置付けられている2)。
JGOG3016試験の結果を受け,米国婦人科腫瘍臨床研究グループ(GOG)によるGOG262試験5),および欧州卵巣がん研究グループ(ICON)によるICON8試験6)が実施された。
GOG262試験5)では,Stage II~IVの上皮性卵巣癌,腹膜癌,卵管癌の患者692例を対象として,dd-TC療法とc-TC療法の有用性が比較された。なお,本試験ではベバシズマブの併用が可能であった(併用率84%)。その結果,dd-TC療法の有用性は確認できず,ベバシズマブ併用時のdd-TC療法は推奨できないものと考えられる。また,ベバシズマブ非併用時についても,dd-TC療法の有用性は不明であると解釈できる。
ICON8試験6)では,Stage Ic~IVの上皮性卵巣癌,腹膜癌,卵管癌の患者1,566例を対象として,c-TC療法群,dd-TC療法群に,dd-TC療法におけるカルボプラチンの投与間隔(3週に1回)を週1回に短縮した(w-TC療法)群を加えた3群において,それぞれの療法の有用性が比較された。なお,本試験ではベバシズマブの併用は行われておらず,JGOG3016試験と近似したプロトコールである。その結果,ベバシズマブ非併用下でのdd-TC療法の有用性は確認できなかった。JGOG3016試験で示されたdd-TC療法の有用性がICON8試験で確認できなかった理由の1つに,人種差があるのではないかといわれている。先述のようにわが国で明細胞腺癌が多数を占めることや1),TC療法を受けた小細胞肺癌患者の日米比較では,一塩基多型(SNP)の相違が治療効果や毒性発現に影響する可能性が示唆されている7)。
分子標的治療薬
1)ベバシズマブ
血管内皮増殖因子(VEGF)に対する抗体薬であるベバシズマブは,わが国で最初に卵巣癌に対する分子標的治療薬として保険収載された。ベバシズマブの初回化学療法のTC療法への上乗せ効果を検討した主な第III相試験には,GOG218試験8)とICON7試験9)がある。
GOG218試験8)では初回腫瘍減量手術後のStage IIIおよびIVの卵巣癌患者1,873例を対象にc-TC療法に加え,ベバシズマブ15mg/kgが21サイクル(c-TC療法2サイクル目~22サイクル目)投与され,c-TC+プラセボ群と比較された。PFS中央値はc-TC+プラセボ群で10.3ヵ月,c-TC+ベバシズマブ群で14.1ヵ月となり,有意な延長が認められた(HR=0.717)。
ICON7試験9)では,Stage I~IVの卵巣癌患者1,528例を対象にc-TC療法に加え,ベバシズマブ7.5mg/kgがc-TC療法に並行して6サイクル,c-TC療法終了後12サイクル投与され,c-TC+プラセボ群と比較された。その結果,PFS中央値はc-TC+プラセボ群で17.3ヵ月,c-TC+ベバシズマブ群で19.0ヵ月となり,有意な延長が認められた(HR=0.81)。
以上のように,初回化学療法においてベバシズマブの上乗せにより,PFS中央値は有意な延長が認められたが,OSの延長は認められなかった。また,現在のところベバシズマブの上乗せによるOS延長を検証した報告は見当たらない。癌治療の目標をOS延長に置くという観点からは,ベバシズマブの上乗せ効果についての評価は定まっていないとみるのが妥当であると考えられる。
2)オラパリブ
Poly(ADP-ribose) polymerase(PARP)阻害薬であるオラパリブは,プラチナ製剤感受性再発卵巣癌に対する維持療法薬として2018年に保険収載された。
Ledermannらによるプラチナ製剤感受性である再発卵巣癌に対するオラパリブ(400mgを1日2回)による維持療法の有用性を検討したランダム化第II相試験では10),直前の化学療法により完全奏効(CR)または部分奏効(PR)を得たhigh grade再発卵巣癌患者265例(オラパリブ群136例,プラセボ群129例)が対象となった。その結果,PFS中央値はオラパリブ群で8.4ヵ月,プラセボ群で4.8ヵ月となりPFSの有意な延長が認められた(HR=0.35)。OS中央値についてはオラパリブ群で29.7ヵ月,プラセボ群で29.9ヵ月となり有意な延長は認められなかった(HR=0.94)。
BRCA変異陽性のプラチナ製剤感受性再発卵巣癌患者を対象に,オラパリブ(300mgを1日2回投与)の有用性を検討した第III相試験であるSOLO 2試験では11),直近の化学療法によりCRまたはPRを得たhigh grade再発卵巣癌患者295例(オラパリブ群196例,プラセボ群99例)が対象となった。その結果,PFS中央値はオラパリブ群で19.1ヵ月,プラセボ群で5.5ヵ月となりPFSの有意な延長が認められた(HR=0.30)。
以上のように,オラパリブはhigh grade再発卵巣癌患者のPFSを有意に延長することが示されたが,OSの延長に寄与するかについては,今後のエビデンスの蓄積が待たれる。
免疫チェックポイント阻害薬
卵巣癌治療において,OS延長効果を示すbreakthrough therapyに関するエビデンスは近年得られていない。このような状況下,免疫チェックポイント阻害薬は,OSの延長が期待できる薬剤として注目される。免疫チェックポイント阻害薬については,化学療法治療抵抗性を示した卵巣癌患者を対象とした,複数の第III相試験が進行中である。
1)抗PD-1抗体
Hamanishiらによるプラチナ抵抗性再発・局所進行性卵巣癌に対する抗programmed cell death 1(PD-1)抗体であるニボルマブの第II相試験では12),ニボルマブ1mg/kgを2週おきに投与するコホート(n=10)および,3mg/kgを2週おきに投与するコホート(n=10)において,その有用性が検討された。その結果,3mg/kgを投与したコホート中の2例がCRを得た。CRを得た患者のうち1例の組織型は,化学療法感受性の低い明細胞癌であった。
Vargaらによるprogrammed cell death ligand 1(PD-L1)を発現している固形癌を対象として,ペムブロリズマブの有用性を検討した第Ib相試験が進行中であるが13),本試験には化学療法抵抗性となった卵巣癌コホート(n=26)が含まれている。その中間解析結果では,卵巣癌コホートにおいて1例がCR,2例がPR,6例が安定(SD)を得た。
2)抗PD-L1抗体
Disisらによる進行性卵巣癌に対するアベルマブの有用性を検討した第Ib相試験では14),124例中12例がPR,55例がSDを得た。また,対象患者中の74例において,PD-L1発現の有無による検討が行われた。その結果,PD-L1陽性患者57例の奏効率は12.3%,PD-L1陰性患者17例の奏効率は5.9%であった。
3)抗CTLA-4抗体
Matulonisらによる顆粒球マクロファージコロニー刺激因子(GM-CSF)の分泌を促進する癌ワクチンで治療された卵巣癌患者11例を対象に,抗cytotoxic T lymphocyte-associated antigen 4(CTLA-4)抗体であるイピリムマブの有用性を検討した第II相試験では15),1例がPR,4例がSDを得た。
4) 免疫チェックポイント阻害薬の期待と課題
免疫チェックポイント阻害薬は,非小細胞肺癌,メラノーマ,腎細胞癌,卵巣癌などの固形癌に対する有用性が期待できる反面,卵巣癌に対する複数の臨床試験結果により,単剤投与の奏効率は6~15%と比較的低いことが示された(表1)。

表1 免疫チェックポイント阻害薬の卵巣癌に対する臨床試験
しかし,癌の治療目標がOS延長である以上,免疫チェックポイント阻害薬によるOS延長のエビデンスは重要であり,たとえ高い奏効率が期待できない場合であっても,免疫チェックポイント阻害薬単剤投与に対する期待は大きいと考えられる。ただし,免疫チェックポイント阻害薬では,奏効の基準となるバイオマーカーが確立されていない点が大きな課題であり,これが奏効率低下の要因の1つとなっている。そこで,高頻度マイクロサテライト不安定性(MSI-High),DNAミスマッチ修復不全(MMR-D)などを含むバイオマーカーの探索が,精力的に進められている。これに関連して,免疫チェックポイント阻害薬に感受性の高い,いわゆる“hot tumor”と,感受性の低い“cold tumor”が存在するという見解もあるが,hot tumorは多くの遺伝子変異を保持しているといわれている。
近年,奏効率の向上などを目的とする免疫チェックポイント阻害薬と他剤の併用あるいは免疫チェックポイント阻害薬同士の併用についても検討されている。Jiaoらによるin vitroの検討では16),培養乳癌細胞にPARP阻害薬を添加したところ,PD-L1の発現が亢進した結果,PARP阻害薬の抗腫瘍効果が減弱したが,PARP阻害薬と抗PD-L1抗体を同時に添加した実験系では,これら2剤の単独投与時に比べ有意な抗腫瘍効果の増強が認められた。Pfirschkeらによる肺腺癌モデルマウスを用いた検討では17),免疫応答を誘導する化学療法薬が,免疫チェックポイント阻害薬の抗腫瘍効果を増強することが示された。このような知見に基づき,免疫チェックポイント阻害薬の単剤投与だけでなく,化学療法薬やPARP阻害薬との併用の有用性を検討する臨床試験が進行中である(表2)18)。
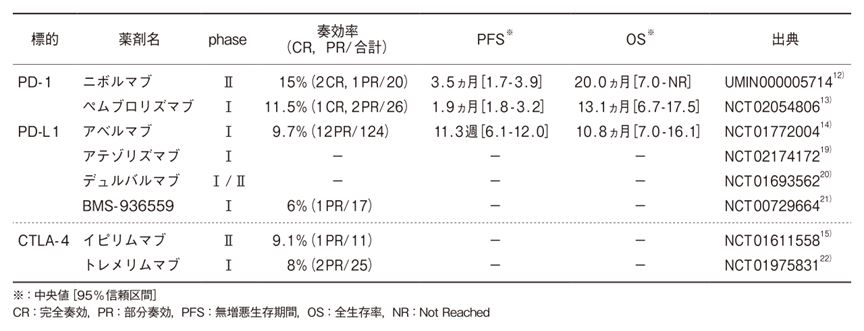
表2 進行中の卵巣癌に対する免疫チェックポイント阻害薬の臨床試験(単剤,併用)
腫瘍内科医の養成が重要
稿を終えるにあたり,腫瘍内科医養成の重要性について述べる。
免疫チェックポイント阻害薬は期待の持てる癌治療薬の1つであるが,新規の機序を有する薬剤であり,今後どのような副作用が発現するのか,副作用への対処をいかにするかについて臨床経験の蓄積が必須である。婦人科癌についても同薬の適応が見込まれているが,臨床経験の蓄積にはかなりの時間を要するだろう。そのため,腫瘍内科医と婦人科医との協働が必要である。
また,癌治療では,エビデンスに基づく治療と,医療者による患者の「こころのケア」が重要である。腫瘍内科医は,抗癌剤治療のエビデンスの把握と「こころのケア」に携わる役割を担っている。一方,わが国の腫瘍内科医数は1,000人を超えたところであるが,協働の重要性などを含めた癌治療の現状を考えると5,000人程度の腫瘍内科医が必要であるといわれている。腫瘍内科医の養成が重要な所以である。
特に婦人科癌の領域では,患者も同性の医師には気軽に相談できる面があり,また,「こころのケア」に必要なnarrativeな対応には,男性に比べ女性に一日の長があると感じている。実際に米国では,すでに腫瘍内科領域は女性医師が多数を占め,海外の関連学会の会場でも女性医師の活躍が目立つ。わが国でも,腫瘍内科領域に多くの女性医師が進出することを強く願っている。
- 日本婦人科腫瘍学会(編). CQ12組織型を考慮した初回化学療法は推奨されるか?. 卵巣がん治療ガイドライン2015年版.金原出版; 東京 2015. p.88-90.
- 日本婦人科腫瘍学会(編). CQ9推奨される初回化学療法のレジメンは?. 卵巣がん治療ガイドライン2015年版. 金原出版;東京 2015. p.78-81.
- Katsumata N, Yasuda M, Takahashi F, et al. Dose-dense paclitaxel once a week in combination with carboplatin every 3 weeks for advanced ovarian cancer: a phase 3, open-label, randomised controlled trial. Lancet. 2009; 374: 1331-8.
- Katsumata N, Yasuda M, Isonishi S, et al. Long-term results of dose-dense paclitaxel and carboplatin versus conventional paclitaxel and carboplatin for treatment of advanced epithelial ovarian, fallopian tube, or primary peritoneal cancer (JGOG 3016) : a randomised, controlled, open-label trial. Lancet Oncol. 2013; 14: 1020-6.
- Chan JK, Brady MF, Penson RT, et al. Weekly vs. Every-3-Week Paclitaxel and Carboplatin for Ovarian Cancer. N Engl J Med. 2016; 374: 738-48.
- Clamp AR, McNeish I, Dean A, et al. ICON 8: A GCIG Phase III randomised trial evaluating weekly dose-dense chemotherapy integration in first-line Epithelial Ovarian/Fallopian Tube/Primary Peritoneal Carcinoma (EOC) treatment: Results of Primary Progression- Free Survival (PFS) analysis. ESMO 2017; Abstract 9290_PR.
- Gandara DR, Kawaguchi T, Crowley J, et al. Japanese-US common-arm analysis of paclitaxel plus carboplatin in advanced non-small-cell lung cancer: a model for assessing population-related pharmacogenomics. J Clin Oncol. 2009; 27: 3540-6.
- Burger RA, Brady MF, Bookman MA, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. 2011; 365: 2473-83.
- Perren TJ, Swart AM, Pfisterer J, et al. A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med. 2011; 365: 2484-96.
- Ledermann J, Harter P, Gourley C, et al. Olaparib maintenance therapy in platinum-sensitive relapsed ovarian cancer. N Engl J Med. 2012; 366: 1382-92.
- Pujade-Lauraine E, Ledermann JA, Selle F, et al. Olaparib tablets as maintenance therapy in patients with platinum-sensitive, relapsed ovarian cancer and a BRCA1/2 mutation (SOLO2/ENGOT-Ov21) : a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2017; 18: 1274-84.
- Hamanishi J, Mandai M, Ikeda T, et al. Safety and Antitumor Activity of Anti-PD-1 Antibody, Nivolumab, in Patients With Platinum-Resistant Ovarian Cancer. J Clin Oncol. 2015; 33: 4015-22.
- Varga A, Piha-Paul SA, Ott PA, et al. Pembrolizumab in patients (pts) with PD-L1–positive (PD-L1⁺) advanced ovarian cancer: Updated analysis of KEYNOTE-028. J Clin Oncol. 2017; 35 (15_suppl) : 5513.
- Disis ML, Patel MR, Pant S, et al. Avelumab (MSB0010718C; anti-PD-L1) in patients with recurrent/refractory ovarian cancer from the JAVELIN Solid Tumor phase Ib trial: Safety and clinical activity. J Clin Oncol. 2016; 34 (15_suppl) : 5533.
- Matulonis UA, Hodi S, Dranoff G, et al. Long-term survival and outcome update on patients (pts) with recurrent ovarian cancer who received Ipilimumab post GVAX vaccine. Clin Cancer Res. 2016; 22 (supplement) : B72.
- Jiao S, Xia W, Yamaguchi H, et al. PARP Inhibitor Upregulates PD-L1 Expression and Enhances Cancer-Associated Immunosuppression. Clin Cancer Res. 2017; 23: 3711-20.
- Pfirschke C, Engblom C, Rickelt S, et al. Immunogenic Chemotherapy Sensitizes Tumors to Checkpoint Blockade Therapy. Immunity. 2016; 44: 343-54.
- Gadducci A, Guerrieri ME. Immune Checkpoint Inhibitors in Gynecological Cancers: Update of Literature and Perspectives of Clinical Research. Anticancer Res. 2017; 37: 5955-65.
- ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/ NCT02174172(2018年11月現在)
- ClinicalTrials.gov. https://clinicaltrials.gov/ct2/show/ NCT01693562(2018年11月現在)
- Brahmer JR, Tykodi SS, Chow LQ, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012; 366: 2455-65.
- Callahan MK, Odunsi K, Sznol M, et al. Phase 1 study to evaluate the safety and tolerability of MEDI4736 (durvalumab, DUR) + tremelimumab (TRE) in patients with advanced solid tumors. J Clin Oncol. 2017; 35 (15_Suppl.) : 3069.

